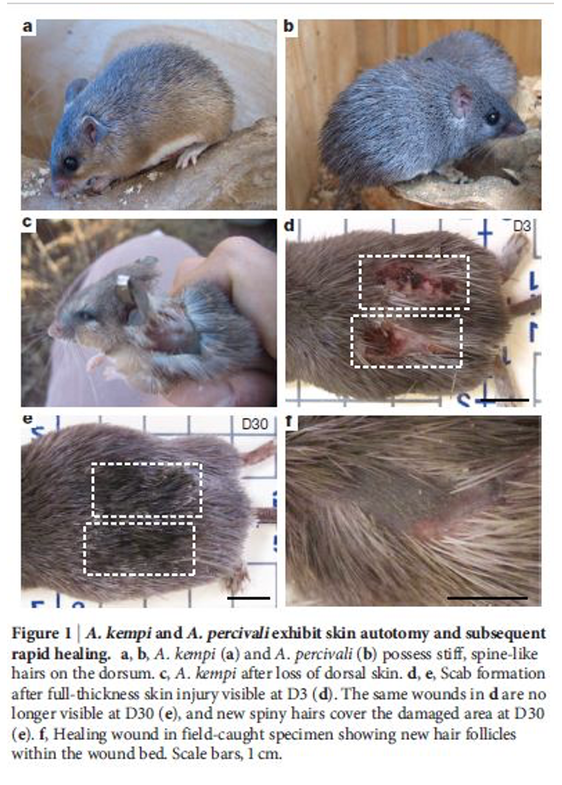
Image by Marcel Burkhard Image by Marcel Burkhard According to the legend, oyster fishermen only, unwittingly, doubled their trouble by cutting and disposing of any starfish they dredged up back into the ocean. Why, because starfish can regenerate their lost limbs. So do salamanders, newts, and many other amphibians who sacrifice a part of their body or entire limb to escape predators. And, that too, without any trace of scar tissue. Regenerative adaptations might seem oddly comical; much like Marvel’s superhero Wolverine, which can regrow its lost tissues and limbs after horrific battles. These biological traits are useful in the real world, particularly in the animal kingdom, where the resources are scarce and competition is intense. It can be quite a good idea to shed your skin or lop off a part of yourself, and then regrow it, rather than be eaten up by your predator. Mammals, however, are not so fortunate. When any of us get injured or lose our body parts, we do so permanently. The only exception here is African spiny mice, Acomys, which can regrow its lost skin and seal small holes in its ears thanks to its unique gene expression. The study of Acomys might provide us with the means to better understand this incredible healing power. Spiny mice and Seifert: A skeptical encounter Spiny mice, native to Africa, the Middle East, and southern Asia, are close relatives of mice and rats. They are aptly named for their spiny hairs that cover their backs much like hedgehogs, albeit not as hard to hurt a predator. Another important morphological feature is their almost bald, brittle tails that break off either as a whole or in part. Acomys is emerging as an important animal model for evolution and development studies, says Cheryl L Haughton. The first one to publish a paper on spiny mice regeneration in Nature, Ashley Seifert of the University of Florida reported that two species of the African spiny mouse; Acomys kempi and Acomys percivali exhibit remarkable abilities of skin shedding and wound regeneration. Intrigued by the anecdotal reports that ecologists are left with nothing but a piece of skin in their hands when they try to catch & trap a spiny mouse, Seifert went to Kenya to see for himself. Seifert tried to capture the aforementioned species of spiny mice in the rocky hills of Kenya where they lived. In mechanical tests, he observed that most of the vigorously moving animals had their skins shed and tails removed. He was surprised to see that the skin of a spiny mouse is 77 times softer, or brittle you could say, than a typical lab mouse. He compared regeneration and scarring as, “Regeneration seems to be an all or nothing trait. I think of them as two extremes on a spectrum. It’s really rare to find an organism that ‘kind of’ regenerates. For example, it’s impossible to find a salamander that regrows only half a limb.” The skin of spiny mice rips off their back so easily when handled, and they also detach their tail frequently. Losing such large chunks of skin would be a disaster for most mammals. Acomys take advantage of this biological trait, autotomy, to get away from the jaws/claws of their predators. Losing some skin is better than losing an entire limb, what do you say? They can also swiftly replace the flaying skin, hair follicles, and fat cells. Unlike humans, Acomys completely replace every layer of their skin, without any scar tissues to seal off the wound. Scarring is unappealing, as the tissues are harder and darker than the surrounding skin. Timeline of regeneration events at cellular levels... The skin of a spiny mouse and a typical lab mouse superficially look-alike, albeit the former is loose and fragile. It takes 77 times more energy to break the skin of a typical house mouse than to tear the skin of an African spiny mouse. At the cellular levels, African spiny mice have far greater proportions of hair follicles and associated glands. As a result, there are fewer connective tissues to hold their skin together. But the scientist found that spiny mice complement their weak skin with super-fast healing. After a few pricks from Seifert and colleagues, the bleeding promptly stopped and the scab reappeared. What an adult mouse would take 5 to 7 days to grow new skin, Acomys did in just 3 days. 10 days after the wound and the skin had healed and renewed completely without scars. Day 21 and mice were growing new hair follicles to replace the ones they had lost. Instead of laying down new collagen fibers in parallel, dense layers, the spiny mice deposit it in a loose woven network, similar to that in healthy skin, resulting in scar-free healing. A typical lab mouse would fail to do so. What’s more intriguing is spiny mice’s ability to heal and regenerate 4-millimeter holes in their ears. They seal off the holes with new skin, regrow hairs, fat cells, cartilage, and muscle, though muscles were not reliably reproduced, without any trace of scar tissue. Epimorphic regeneration The process of regeneration with which salamanders and newts regrow an entire new limb is known as epimorphic regeneration. One of the most important steps of the process is blastema formation. The blastema is a mass of dividing cells that revert to an immature, undifferentiated embryonic-stem-like cell from which different kinds of tissues arise and grow into new limbs. Seifert observed a clump of undifferentiated cells, much like blastema, around the ear wound in a spiny mouse. “It looked and behaved like a mammalian blastema!”, said Seifert & colleagues. The blastema preserves its regenerative state, all credits to signals from the “wound epidermis”, a specialized skin layer that forms above it. If either of the two fails to form, the lost limb won’t grow back. Scientists found that spiny mice heal their ear wounds similarly. When holes are punched in their ears, they immediately form a mass of cells; the molecular hallmarks of a salamander’s blastema. Dr. Seifert believes that Acomys possess inactive genes that prevent regeneration in humans. Later, a group of scientists from the Wistar Institute, Philadelphia showed that the reason Acomys can repair earholes is not that they have a mutant gene or an extra gene, rather the lack of gene p21 expression. If a common lab mouse has an inactive p21 gene, the healing process can go as far as healing ear holes completely. The studies confirm that mammalian regeneration is not as out-of-reach as once was thought. P21 and other genes suppress the regenerative abilities of most mammals. A major setback to human regeneration is the formation and maintenance of blastema. It would take extended research on these stars of regeneration like salamanders and spiny mice to generate therapeutic treatments for humans. If only our regenerative abilities are enhanced, controlling these genes, will open the prospects of healing wounds in humans without scarring. And the work on African spiny mice, non-traditional models for human regeneration, is extremely valuable for scientists who, now, are looking for new opportunities to investigate mechanisms of tissue regeneration in mammals. AuthorNida Riaz is a freelance blogger based in Pakistan. She started writing about her passion for the environment when the world came to a stop in early 2020.
0 Comments
Leave a Reply. |
|
|
(833) CMS-LINE
(833) 267-5463 PO Box 13477 Mill Creek, Wa, 98082 © Conservation Made Simple. All rights reserved.
501(c)(3) Non-Profit, Tax ID#: 82-1646340 Copyright © 2021 Conservation Made Simple |